EATRIS Coordinator in Latvia
A coordination center in Latvia that promotes the development of translational medicine by providing researchers with access to international research infrastructure, necessary resources, and strategic partnerships.


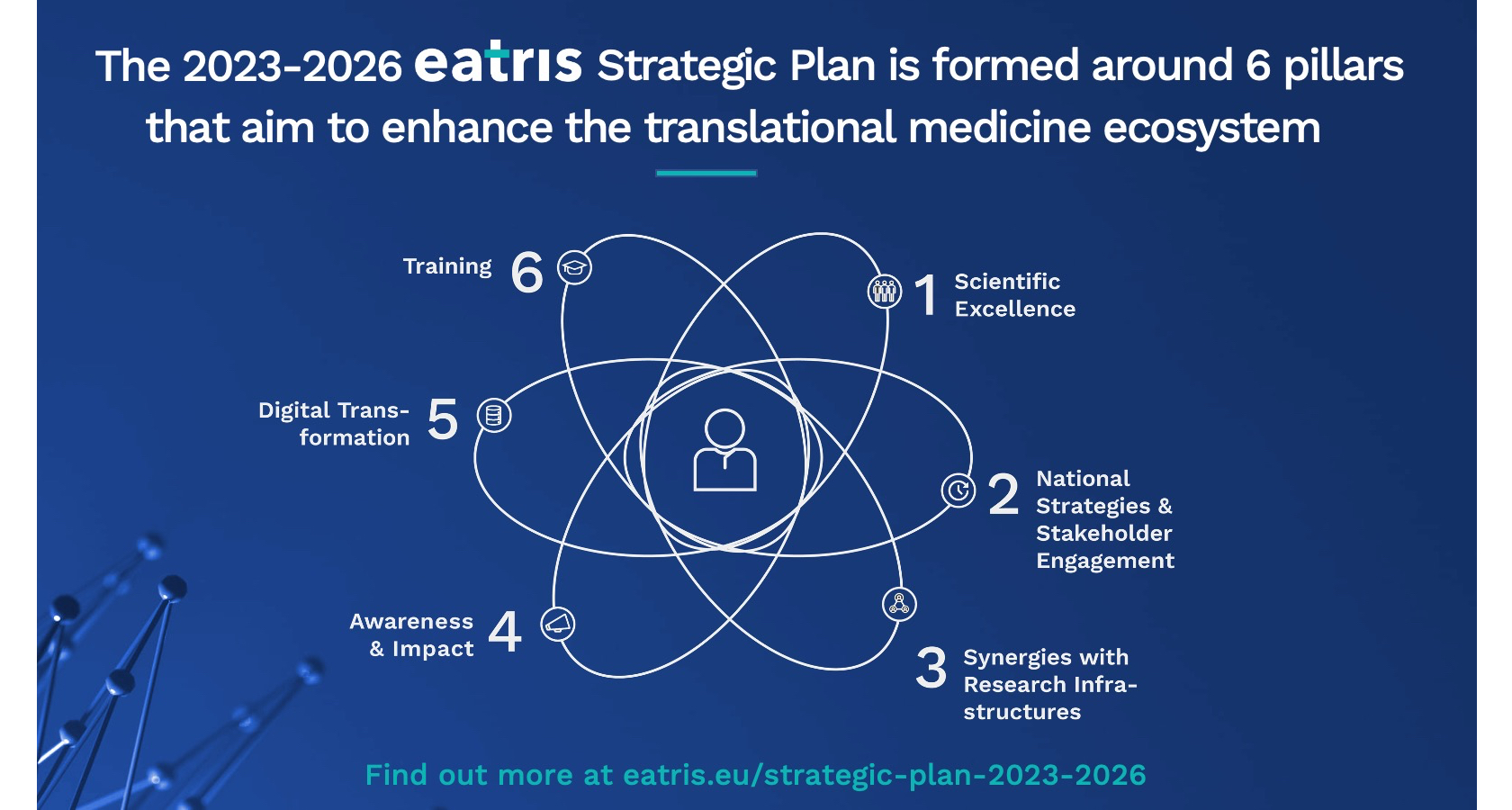
EATRIS is a non-profit European Research Infrastructure Consortium (ERIC). This specific legal form is designed to facilitate the joint establishment and operation of research infrastructures of European interest.
The organisational model of EATRIS is based on the unification of European countries with the aim of promoting the development and scientific excellence of translational medicine, the development of new diagnostic methods and drugs and ensuring their implementation in practical medicine in accordance with ethical and regulatory requirements.
Product platforms
- Advanced therapy medical products
- Biomarkers
- Imaging & Tracing
- Small Molecules
- Vaccines
- COVID-19 research
EATRIS has published an update on their response to the COVID-19 pandemic, which also includes an overview of our institutions' activities and services.
Vision
To make the translation of scientific discoveries into medical products more effective in order to improve health and quality of life.
Mission
To support researchers in developing their biomedical discoveries into novel translational tools and interventions for better societal health outcomes.
Aim
To help scientists with the resources they wouldn’t be able to have otherwise, like laboratory equipment, guidance, or other scientific specialties.
Supporting academia, industry, patients and policy makers
EATRIS makes the translational community accessible to researchers by providing:
- Access to quality research infrastructure services;
- Support for multi-disciplinary, patient-oriented research;
- The best infrastructure & resources to ensure the optimal methodological approach;
- Easy access to industry, regulatory expertise;
- The development of new tools for better R&D output.
EATRIS assists in the preparation of project applications:
- By establishing a consortium
- Joining as partner
- Acting as presenter.
More on the EATRIS Consortium, its objectives, tasks and opportunities
News
EATRIS Strategic Plan 2023–2026
EATRIS+

A Flagship Initiative in Personalised Medicine
EATRIS-Plus project aims to support the long-term sustainability of the European Research Infrastructure for Translational Medicine (EATRIS) by delivering innovative scientific tools to the research community, strengthening the EATRIS financial model, and reinforcing EATRIS’ leadership in the European Research Area in the field of Personalised Medicine (PM).
Project in a Nutshell
|
Mission Oriented Towards Patients
PM research seeks to identify interventions that can be targeted to individual patients based on their predicted response. This has enormous value for patients, as it can reduce trial-and-error treatments, enable better choices for determining which medications would be most effective, which in turn can help to manage rising healthcare costs. EATRIS-Plus will work towards improving the understanding, data-sharing, and clinical needs of how gene, protein, mRNA and metabolite analysis will directly impact PM solutions.
Scientific Rationale
Efficient advancement of PM depends on the availability of validated patient-targeted biomarkers. However, we need a more precise understanding of the molecular profiles of individuals, which is facilitated by multi-omic approaches. In order to turn the multi-omic promises into a reality, systemic bottlenecks impacting the biomarker field need to be overcome.
The consortium ambition is to deliver a multi-omic toolbox to support cross-omic analysis and data integration in clinical samples. The omic tools will be developed and tested with a real-world setting healthy demonstrator cohort in Czechia. Information available on this cohort will be augmented during the project with transcriptomic, proteomic and metabolomic data. By the end of the project the multi-omic toolbox will be made available to the research community.
Capacity Building and Long-Term Sustainability
Highlights of EATRIS-Plus
|
EATRIS-Plus is a comprehensive sustainability project that involves strategic stakeholder engagement, with the aim to increase EATRIS industry and international collaborations, as well as provide training opportunities to the EATRIS research community and beyond. The long-term sustainability of EATRIS will be supported by increased efforts in node capacity-building and outreach, the launch of “EATRIS Quality Certificate”, the onboarding of at least 4 new countries by 2023 and the establishment of further Open Innovation Hubs with industry partners. Among the most important training initiatives the further development of the free-of-charge e-learning environment TransMed Academy, the organisation of 3 Personalised Medicine Summer Schools, and development of online training for patients.
EATRIS is a non-profit European Research Infrastructure Consortium (ERIC) providing research services, laboratory and clinical expertise, and high-end technologies in translational medicine.
![]()
Workshops and courses
Free online course, Introduction to Translational Research for Rare Diseases, where you will gain an overview of the issues, challenges, and opportunities in translating research findings into treatments for rare diseases.

- Past events
Workshop on ATMPs Development in Rome, 20–24 June 2022
Workshop on the Theory and Practice of Industry and Academia Collaboration
- 14 October 2021– Euro-BioImaging user forum: Understanding and Fighting Cancer
- 21 & 28 October, 4 November 2021 – Drug repurposing for rare diseases workshop series
- 25 October 2021 – The impact of COVID-19 on routine vaccination
- 5 November 2021 – EATRIS Horizon Funding Webinars: Building meaningful patient engagement in your research proposal
- 26 November 2021 – EATRIS Horizon Funding Webinars: How to strengthen research quality practices in your grant application?
- 29 November 2021 – EMA (European Medicines Agency) and EATRIS present: navigating the regulatory requirements for ATMPS (advanced therapies)
More information about these and other events organised by EATRIS
Translation Together
Translation Together is a unique collaboration of leading translational research organisations from around the world leveraging our complementary scientific and operational strengths, our shared insight of the challenges facing translation, and our collective voice to advance the science and understanding of biomedical translation.
Partners
- AdMare Bioinnovations
- LifeArc
- National Center for Advancing Translational Sciences (NCATS)
- Therapeutic Innovation Australia
- Fiocruz – The Oswaldo Cruz Foundation
- Japan Agency for Research and Development (AMED)
- EATRIS-ERIC

Mission
Translation Together envisions a global translational research community of diverse stakeholders empowered to effectively and efficiently translate discoveries into health interventions for the benefit of patients and society.
Vision
Advancing the science and understanding of biomedical translation through global collaboration.
Objectives and Activities
https://translationtogether.org/activities/
Education & Training
https://translationtogether.org/activities/education-training/
Coordinate and develop programs and resources for educating and training the next generation of translational scientists and other key stakeholders.
Programmes and resources for educating and training from our partners
- adMare
- EATRIS
- FioCruz
- LifeArc
- NCATS
- TIA
Advocacy & Awareness
Advocate for a broad understanding of and appreciation for translation and translational science among diverse stakeholders.
- Check out our article on: The Fundamental Characteristics of a Translational Scientist published in ACS Pharmacol. Transl. Sci.
- Check out our review on Putting Translational Science onto a Global Stage published in Nature Reviews Drug Discovery, where we highlight our initial strategy to reduce or remove bottlenecks in translatioN
- Explore the new journal Translational and Regulatory Sciences (TRS) that our AMED partner created to provide a platform where industry, academic and government experts can harmonize their efforts on equal footing and promote academic progress and dissemination regarding the regulatory science of pharmaceuticals, medical devices and related aspects
Resources & Tools
Assist investigators in the conduct of translation and translational science by connecting them to resources, tools, technologies, and expertise.
- The Assay Guidance Manual by our NCATS partner. The manual is a free, best-practices online resource devoted to the successful development of robust, early-stage drug discovery assays. They also host periodic, free workshops on the Assay Guidance Manuals, including an online workshop.
- Drug Discovery, Development and Deployment Maps by our NCATS partner that provides dynamic and interactive representations of the modern therapeutic development process.
- The NCATS COVID-19 OpenData Portal designed to rapidly and openly share SARS-CoV-2 screening data and assay information with the global health community.
Collaborative Research
Conduct collaborative research projects to remove systemic barriers and catalyze translation.
https://translationtogether.org/activities/collaborative-research/
TRANSMED Academy
To support biomedical researchers gain a better understanding of translational research and medicine development, EATRIS has developed its very own e-learning platform that offers access to several online activities free of charge. Online training modules are a great way to provide high quality, accessible and interactive training at all levels.
TRANSMED Academy offers three types of e-learning content:
- Introductory modules for early career researchers and expert patients;
- Targeted modules on the use of specific translational technologies for academics and SME based researchers developing Personalised Medicine research;
- Modules and recorded webinars on cross-cutting topics related to funding, impact or technology transfer for the research community at large.
https://eatris.eu/solutions/transmedacademy/
Seminar 'Translational medicine – from molecule to the patient'
Reports
EATRIS 2022 Annual Report | Dedicated website
EATRIS 2021 Annual Report | Dedicated website
Contact
National hub coordinator
Lead Researcher, Institute of Microbiology and Virology